Chitosan Derivatives - Promising Sustainable Biopolymers for Quality Healthcare Textiles
Introduction
The consumers are now increasingly aware of the hygienic lifestyle and there is necessity and expectation for high quality healthcare textiles. That’s why it is one of the most important, continuously expanding and growing fields in technical textiles. Healthcare textiles represent structurally designed and accomplished for a new health and hygiene-related textiles products for the well-being of mankind which applications is diverse, ranging from hospitals, hotels to personal care products. Because of their importance, a number of chemicals have been employed to impart antimicrobial activity to textile materials. These chemicals include inorganic salts, organometallics, phenols and thiophenols, onium salts, antibiotics, heterocyclic compounds with anionic groups, nitro compounds, urea and related compounds, formaldehyde derivatives, amines and synthetic dyes [1]. However, with the public’s enhanced awareness of eco-safety, there has been considerable debate about their use, because majority of such agents are toxic to humans and are not environmentally friendly. In addition, another big concern is that some of these agents are being increasingly resisted by microbial pathogens. Therefore, the role of textile finishers has now become increasingly demanding and has strengthened the interest in alternative ecofriendly and biodegradable finishing agents [2-5].
Chitosan is the deacetylated derivative of chitin that is the second most abundant polysaccharide found on earth next to cellulose. When chitin is deacetylated over about 60% it becomes soluble in dilute aqueous acids and is referred to as chitosan. Chitin is the main component in the shells of crustaceans, such as shrimp, crab [6-9]. Huge amounts of crab and shrimp (prawn) shells have been abandoned as wastes by worldwide seafood companies. This has led to considerable scientific and technological interest in chitin and chitosan as an attempt to utilize these renewable wastes. The applications of chitosan include uses in a variety of areas, such as pharmaceutical and medical applications, paper production, textile dyeing and finishing, fiber formation, wastewater treatment, biotechnology, cosmetics, food processing, and agriculture [10,11]. There is a greater demand for antimicrobial finishes on textile goods. Chitosan, a natural biopolymer, has many chemical attributes, especially its cationic nature, to make it an interesting candidate for these applications. However, the major problems of chitosan are its loss of the antimicrobial activity under alkaline conditions due to its loss of the cationic nature. But chitosan derivatives will overcome that problem due to retain antimicrobial activity in wide range of pH that could have a strong economic, social, and environmental impact, especially in our country.
In view of these ecological, environmental concerns and to overcome the drawback of chitosan, we are going to explore chitosan derivatives which will in turn help to develop next generation healthcare textiles. This will be followed by a focus on some recent developmental works pertaining to antimicrobial finishing of textiles using various “green chemistry” approaches in order to provide safe and novel antimicrobial textiles for aesthetic, hygienic, and healthcare applications in the near future. The specific objectives of the present project are: preparation of sustainable chitosan derivatives from prawn shell waste, modification of cotton fabric with chitosan derivative and antimicrobial assessment and different characterization of modified cotton fabric as for healthcare textiles. The prepared chitosan and its derivatives will be applied on cotton fabric and the modification will be confirmed using FTIR, TGA and DTA techniques. As quality healthcare textile, antimicrobial test, water vapor transmission rate, swelling test, and tensile strength of finished and unfinished cotton fabric will be investigated.
Experimental
Materials
Shrimp shell was collected from Mongla (near Sundarban forest), Khulna, Bangladesh that are waste of shrimp processing area. Cotton fabrics were collected from Keya spinning mills Ltd., Bangladesh. Escherichia coli and Staphylococcus aureus bacteria are collected from “Biochemistry and Molecular Biology” department of Rajshahi University. All chemicals that were used in the present investigation were of reagent grade.
Methods
Processing of Prawn Shell Waste: Prawn shell was collected, washed, dried, and ground to 40-60 mesh using a hammer mill. The ground prawn shell was then ready for a series of chemical treatment for extraction of chitin. Through a set of chemical treatments demineralization, de-protenization and decolouration of chitin was obtained. Later deacetylation of chitin gave chitosan [12,13].
Preparation of TMC: TMC was synthesized through nucleophilic substitution with CH3I as a reagent, sodium iodide as catalyst and sodium hydroxide (NaOH) as base with N-methyl pyrrolidone (NMP) as solvent [14].
Preparation of CMTMCC: CMTMCC was synthesized by reductive carboxymethylation of TMCC that was accomplished by a chemical reaction between TMCC and monochloroacetic acid in the presence of sodium hydroxide. An amount of 0.1 g of dried TMCC was suspended in 10 mL of NMP or isopropanol as solvent and left to stir overnight at 45°C. After 20 min, under continuous stirring, 0.5 mL of 50% NaOH was slowly added to the reaction mixture that was stirred for another 45 min. Subsequently, 0.12 g of monochloroacetate (molar ratio 1:3 to TMC) was added for about 25 min and reaction was left for 3 h at 60 °C under a nitrogen atmosphere in order to avoid TMC degradation. Afterwards, pH was adjusted to neutral with 1M HCl followed by filtration, washing with 70% ethanol and subsequently with anhydrous ethanol. CMTMC precipitate was dried under vacuum, dissolved in distilled water and dialyzed for 3 days against water, changing dialysis medium twice per day. The dialyzed CMTMCC was lyophilized and later analyzed [15].
Results and Discussion
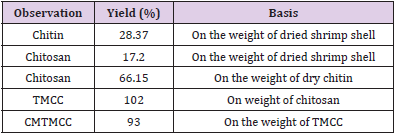
Yield Percentage of Chitin, Chitosan and its Derivatives
The percentage yield was calculated from the ratio of the dried raw sample and the weight of obtained product. This obtained percentage yield is shown in Table 1. The yield percentage of chitosan, TMCC and CMTMCC was 66.5, 102 and 93 % respectively.
FTIR Analysis
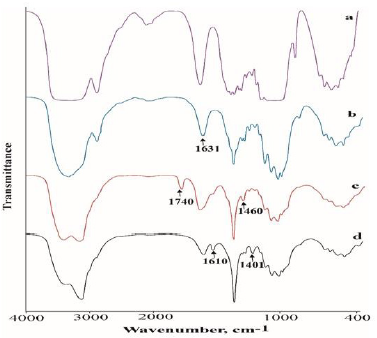
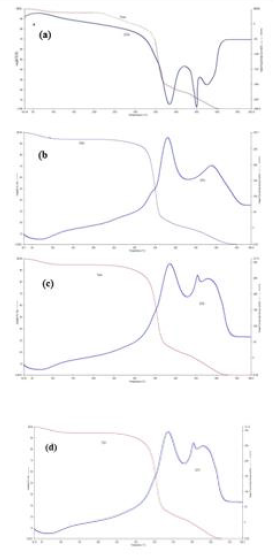
In the FTIR spectrum of the prepared chitosan shown in Figure 1a, there were two absorption peaks at 1631 cm-1 and 2919 cm-1, which correspond to the N−H bending of the primary amine, and the presence of hydroxyl groups respectively. Obvious changes of the FTIR spectra are observed after quaternization of chitosan with methyliodide. In the FTIR spectrum of TMCC, a characteristic peak at 1460 cm-1 indicates the C-H bending of trimethyl ammonium group thus shows evidence of the quaternary ammonium salt group. It should be also noted that the N−H bending (1630 cm-1) of the primary amine disappeared due to the change of the primary amine to the secondary aliphatic amine [16]. A new peak at 1740 cm-1 was also attributed to the quaternary ammonium in TMCC which is shown in Figure 1b. In addition, the spectrum shows a broad band at around 3400 cm 1, probably due to the increased number of hydroxyl groups. The FTIR spectrum of the CMTMCC is shown in Figure 1c. The characteristic peaks of amine (N-H) vibration appeared at 1594 cm-1 for chitosan. Two new absorption peaks at 1401 cm-1 and 1610 cm-1 were attributed to the methyl group of ammonium and carbonyl group in CMTMCC.
The profiles of FTIR spectroscopy of the chitosan, TMCC and CMTMCC synthesized from chitosan are almost similar but have different characteristic peak which shows evidence of the conversion of chitosan to TMCC and CMTMCC.The FTIR spectra of unmodified, chitosan, TMCC modified and CMTMCC modified cotton fabrics were shown in Figure 2. The obtained spectra of both types of fabrics were mostly similar except the new additional peak in the modified cotton fabrics. In case of chitosan modified cotton fabric a new peak at 1631 cm-1 was appeared due to the N-H bending of primary amine. Similarly, characteristic peaks at 1460 cm-1 (C-H bending of trimethyl ammonium group) and 1610 cm-1 (carbonyl group) were attributed to TMCC and CMTMCC modified cotton fabric.
Figure 2: The FTIR spectra of, (a) Unmodified; (b) Chitosan modified; (c) TMCC modified; (d) CMTMCC modified cotton fabric.
Surface Morphology
Surface morphology of the unmodified and modified cotton fabric were evaluated using three dimensional images getting from scanning electron microscope Figure 3. Unmodified cotton fabric exhibits regular surface as well as no adherence. Besides modified fabrics were overlaid with mentioned modifier which reveals little bit irregular, or microstructure roughness helps to increase surface area as well as antimicrobial functionality [17]. Conglomerated granule was evidently visible on the fiber surface which proofs the attachment of applied modifier on the fabric.
Figure 3: The SEM images of, (a) Unmodified; (b) Chitosan modified; (c) TMCC modified; (d) CMTMCC modified cotton fabric.
Antimicrobial Activity
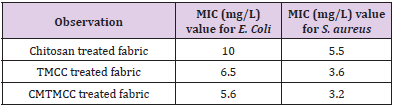
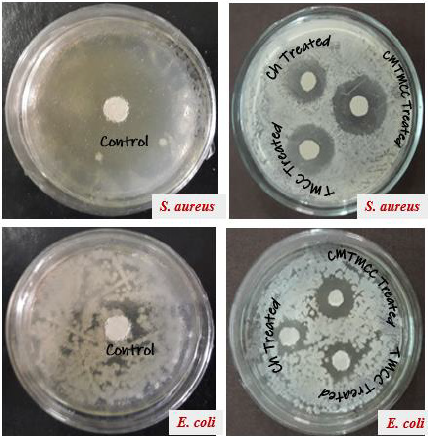
Minimum Inhibition Concentration (MIC) describes the minimum concentration of the modifier in which concentration the multiplication of the bacteria is inhibited. For being different in structure, different modifiers have variation in antimicrobial activity. The Table 2 & Figure 4 show that antimicrobial activity of chitosan, TMCC and CMTMCC treated modified fabric. Chitosan have antimicrobial activity against both E. coli and S. aureus, because of the positive charge on the C-2 of the glucosamine monomer below pH 6. The exact mechanism of the antimicrobial action of chitosan, and their derivatives is still unknown, but different mechanisms have been proposed. Interaction between positively charged chitosan molecules and negatively charged microbial cell membranes leads to the leakage of proteinaceous and other intracellular constituents. Chitosan acted mainly on the outer surface of the bacteria. At a lower concentration the polycationic chitosan does probably bind to the negatively charged bacterial surface to cause agglutination, while at higher concentrations, the larger number of positive charges may have imparted a net positive charge to the bacterial surfaces to keep them in suspension [18,19]. Chitosan interacts with the membrane of the cell to alter cell permeability. But the problem is that it has lower solubility above pH 6 and also its lower attachment capacity on cotton fabric [20]. For TMCC, the mechanism of antimicrobial activity using on cotton fabric is almost similar to chitosan. The slight difference in antimicrobial activity of chitosan and TMCC is due to having positive charge in nitrogen atom and its higher solubility in water than chitosan.
Figure 4: Antimicrobial activity of unmodified and modified cotton fabric against S. aureus and E. coli stain.
The antimicrobial action is believed to occur when the compounds are absorbed onto the bacterial cell surface, increasing the permeability of the lipid cell membrane and causing death through the loss of essential cell materials. In addition, these derivatives of chitosan are generally more active against grampositive and gram-negative bacteria than chitosan. This effect is believed to be due to adsorption of the polymers onto the bacterial cell surface and membrane with subsequent disruption of membrane integrity. Antimicrobial activity generally increases as the content of the quaternary ammonium moiety increases comparatively than chitosan. CMTMCC has better antimicrobial activity than chitosan and TMCC. Here the mechanism is approximately same to them but the difference is that the attachment capacity of CMTMCC (MIC=5.8mg/L for E. coli and 3.3 mg/L for S. aureus) is more than chitosan and TMCC on the cotton fabric.
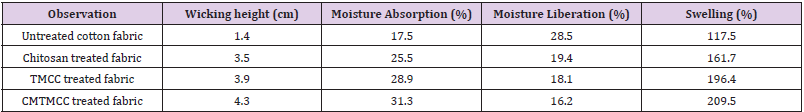
Wicking, Moisture Absorption, Moisture Liberation and Swelling Test Result
Wicking Test: Wicking is the spontaneous flow of a liquid in a porous substrate, driven by capillary forces. Because capillary forces are caused by wetting, wicking is a result of spontaneous wetting in a capillary system. The wicking test results for untreated cotton fabric, chitosan, TMCC and CMTMCC modified cotton are given in Table 3. Untreated cotton fabric shows lower wicking height than the treated cotton fabrics due to the absence of the coating of chitosan, TMCC, and CMTMCC having channel like structure on cotton fabric. Chitosan and its derivatives show higher wicking height (cm). TMCC and CMTMCC have also higher wicking height (cm) comparatively than chitosan treated fabric. The reason for this is TMCC and CMTMCC have channel like structure same as chitosan and also have the presence of positive charge which helps to attract water molecules.
Table 3: Wicking, moisture absorption, moisture liberation and swelling test result of unmodified and modified cotton fabric.
Moisture Absorption: The properties of cotton fiber are strongly affected by moisture content because it is a hygroscopic fiber and absorbs or desorbs moisture from the surrounding atmosphere. In general, the fibers that absorb the greatest amount of moisture are the ones whose properties change the most. The main types of properties affected are: dimensional, mechanical and electrical. The molecular structure of the cotton fiber includes hydroxyl groups that attract water molecules and hydrogen bond with the cellulose [21]. Ability to retain a liquid in the fabric is known as absorption. Absorption consists of several parts, first the fiber surface is wetted and the liquid is transported into the voids between the fibers and are absorbed into the fibers and diffuse [22]. The moisture absorption test results are given in Table 3. The rate of moisture absorption in untreated fabric sample decreased as compared to the treated fabric due to the absence of channel like structure. On the other hand, chitosan, TMCC, and CMTMCC treated cotton fabrics show more moisture absorption (%) due to the presence of hydroxyl groups (-OH) and channel like structure. Also for having the good attachment capacity on cotton fabric CMTMCC treated cotton fabric showed higher moisture absorption (%).
Moisture Liberation: The moisture liberation ability is one of the crucial factors for wounds healing, because wound dressing manages the moisture on the surface and bed of the wound by absorbing and releasing exudates. The Moisture liberation test results of untreated and treated cotton fabrics are given in Table 3. Untreated fabric can easily liberate moisture because of having low capillaries. On the other hand, chitosan, TMCC, and CMTMCC treated cotton fabrics have channel like structure and for this it is unable to moisture liberation.
Swelling Test: Moisture/liquid transport in textile fabrics is one of the critical factors affecting physiological comfort. In conditions where wearers sweat a lot (e.g. high-level bodily activity), it is not only desirable for the fabric next to the skin to absorb liquid rapidly but also to transport it through the fabric promptly to avoid the discomfort of the fabric sticking to the skin. The comfort afforded by textile fabrics can be improved by understanding the liquid transport mechanism [23]. The swelling capacity of an antibacterial fabric plays an important role in the antibacterial activity, wound healing capacity, and for biomedical application due to their high water/solvent holding capacity. They can further absorb a slight to moderate amount of the wound exudates by swelling which helps in fast healing of the wound. The swelling test results are list in Table 3. Swelling % of untreated fabric is lower than others because of lacking the cross-linked channel structure to hold water for being swollen. Then the swelling capacity of CMTMCC is more than others for having not only the presence of hydrophilic groups in the film networks - as like as chitosan and TMCC which assist in improving the swelling characteristic of the fabric but also N atom containing positive charge and higher attachment capacity to the fabric.
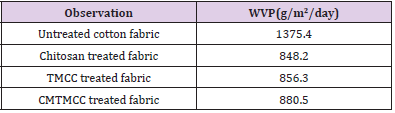
Water Vapor Permeability (WVP) Test
Bolton studied a variety of dressings and determined that a WVTR of about 840 g/m2/day is required to maintain a moist wound surface [24]. Water Vapor permeability occurs mainly for having different vapor pressure on both sides of the cotton fabrics. The WVP test results are given in Table 4. WVP of untreated cotton fabric is high because covering of fabrics with modifiers isn’t occurred on it. For Chitosan, TMCC, and CMTMCC treated fabrics WVP comparatively increases due to the presence of hydroxyl groups. Here after applying Chitosan, TMCC, and CMTMCC on cotton fabric hydrophilic properties are raised and channel like structures are formed which attract water vapor and help to pass away to the outer side of the fabric to saturate the outer side. Among of them CMTMCC have higher WVP because it further has higher attachment capacity on cotton fabric causes more water vapor permeability through cotton fabrics. Fabrics used for wound dressing should have neither higher nor lower WVP value. So for this CMTMCC treated cotton fabrics are more suitable to provide moist wound condition.
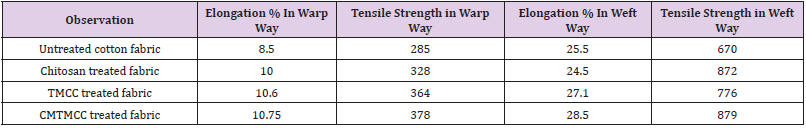
Tensile Strength Test
The tensile strength is one of the most important physical properties of the fabrics. The tensile strength of the fabric contributes greatly to its quality as well as its life. The strength of the fabrics depends on of many different factors that affect it. Molecular structure, temperature, composition, are some of the factors that are responsible for the increase or decrease in the tensile strength of fabric. It is well known that chitosan significantly imparts stiffness in fabric and affect tensile strength of fabric. In this study cotton fabrics are bio-scoured in presence of cellulase enzyme and for this hydrolysis is occurred which causes the loss of tensile strength of cotton fabric. Then to regain the tensile strength the cotton fabrics are treated with chitosan and its derivatives. Tensile strength test results are listed in Table 5. Tensile strength of untreated fabric is low due to hydrolysis during bio-scouring with cellulase. For the chitosan, TMCC, and CMTMCC treated cotton fabrics, tensile strength increases because of binding between fibres and yarns. Chitosan, TMCC, and CMTMCC to bind fibres and yarns and increases tensile strength. Among the samples, CMTMCC treated fabric have higher tensile strength than others.
Thermal Analysis
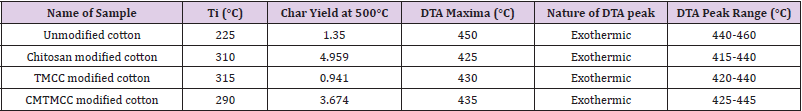
Figure 5 shows TGA and DTA curves of unmodified-, chitosan modified-, TMCC modified- and CMTMCC modified cotton fabrics. It can be seen that the initial decomposition temperature of unmodified cotton fabric is 225OC as shown in Table 6. Also, for DSC curve it is observed that there is a exothermic peak at 450°C which corresponds the burning of the char present in cotton fabric during oxidation. Figure 5b represents TGA and DTA curve for chitosan treated cotton fabric, from this Figure it is obtained two exothermic peaks are seen which correspond to the burning of the incorporated hydrogens and the burning of the char of cotton fabric. The initial decomposition temperature of chitosan cotton fabric is 310OC as shown in Table 6. Now from Figure 5c it is obtained that at 50°C phase transition is occurred, at 80°Can endothermic peak is appeared due to melting behavior and at 340°C another endothermic peak is occurred for the same at 50°C. In this Figure the Initial decomposition temperature was 315°C. And Initial decomposition temperature of CMTMCC modified cotton was 290°C as shown in Table 6.
Figure 5: TGA and DTA curves of, (a) Unmodified cotton; (b) Chitosan modified cotton; (c) TMCC modified cotton; (d) CMTMCC modified cotton.
Conclusion
Different experimental methods were established that chitosan, TMCC and CMTMCC were successfully attached on cotton fabric by pad-dry-cure method. Among the samples, CMTMCC treated fabric have higher tensile strength than others. The rate of moisture absorption in untreated fabric sample decreased as compared to the treated fabric. Antimicrobial activity is the main factor for medical and health care textiles. In this study, chitosan derivatives TMCC and CMTMCC treated cotton fabric exhibited excellent antimicrobial performance. It is expected that this sustainable chitosan derivatives-based healthcare textile retaining durable antimicrobial activities that will be used in hospitals, hotels and personal cares textiles as not only fashion and comfort but also safeguard for human health without disturbing environment. At the same time, the project will help to utilize the by-product or wastage of prawn processing industries.
For more Articles on: https://biomedres01.blogspot.com/



No comments:
Post a Comment
Note: Only a member of this blog may post a comment.